- Smear Preparation
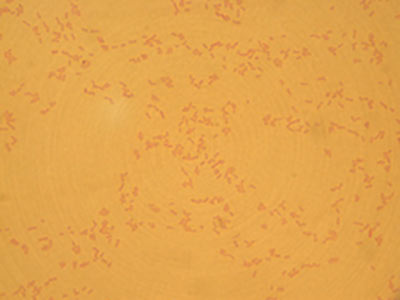
- Simple Stain
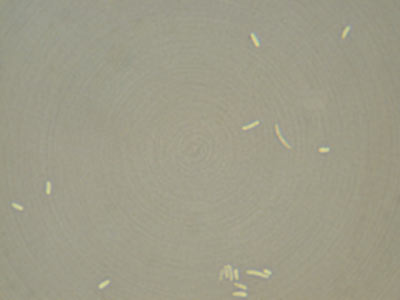
- Negative Stain
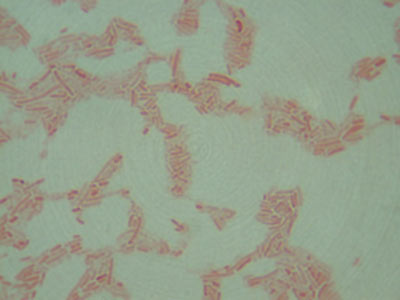
- Gram Stain
- Congo Red Capsule Stain
- Wirtz's Endospore Stain
Smear Preparation
Not only are most bacteria very small, they are also very clear and difficult to view under a microscope without first staining. You must firmly attach your bacteria to a glass slide before you can stain them. There are two important things to consider when preparing a slide for staining:
- The bacteria must be evenly and lightly dispersed. If there are too many bacteria on the slide they will form a big glob and you will not be able to see the morphology of the individual cells. Large blobs of cells also do not stain properly and could yield erroneous results from the improper staining.
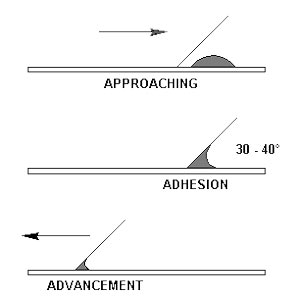
- The bacteria need to be firmly attached to the slide so they are not washed off during the staining procedures. All procedures that attach the bacteria to the slide result in some morphological changes. The cells typically shrink in size and will exhibit some changes in shape and extra-cellular matrices.
You will be preparing slides for staining from both broth and agar surfaces. While the goals are the same for both, evenly and lightly dispersed cells firmly adhered to the slide surface, the techniques are slightly different. Staining is as much art as science. It will undoubtedly take you several tries before you are successful.
Materials
- Clean glass slides
- Inoculating loops or needles
- Sterile water
- Marking pen
- Assorted broth and plate cultures